 Presidency | South Africa □□ November 11, 2020 President #Ramaphosa: In recognition of our country’s manufacturing capability one of the leading pharmaceutical companies in the world, Johnson & Johnson, has entered into an agreement with a local company, Aspen Pharmacare, to manufacture and package its candidate vaccine. 
The vaccine only needed one dose, it didn’t need ultra-cold storage and thus seemed to be the most “African-friendly vaccine”, said Nicolaou. 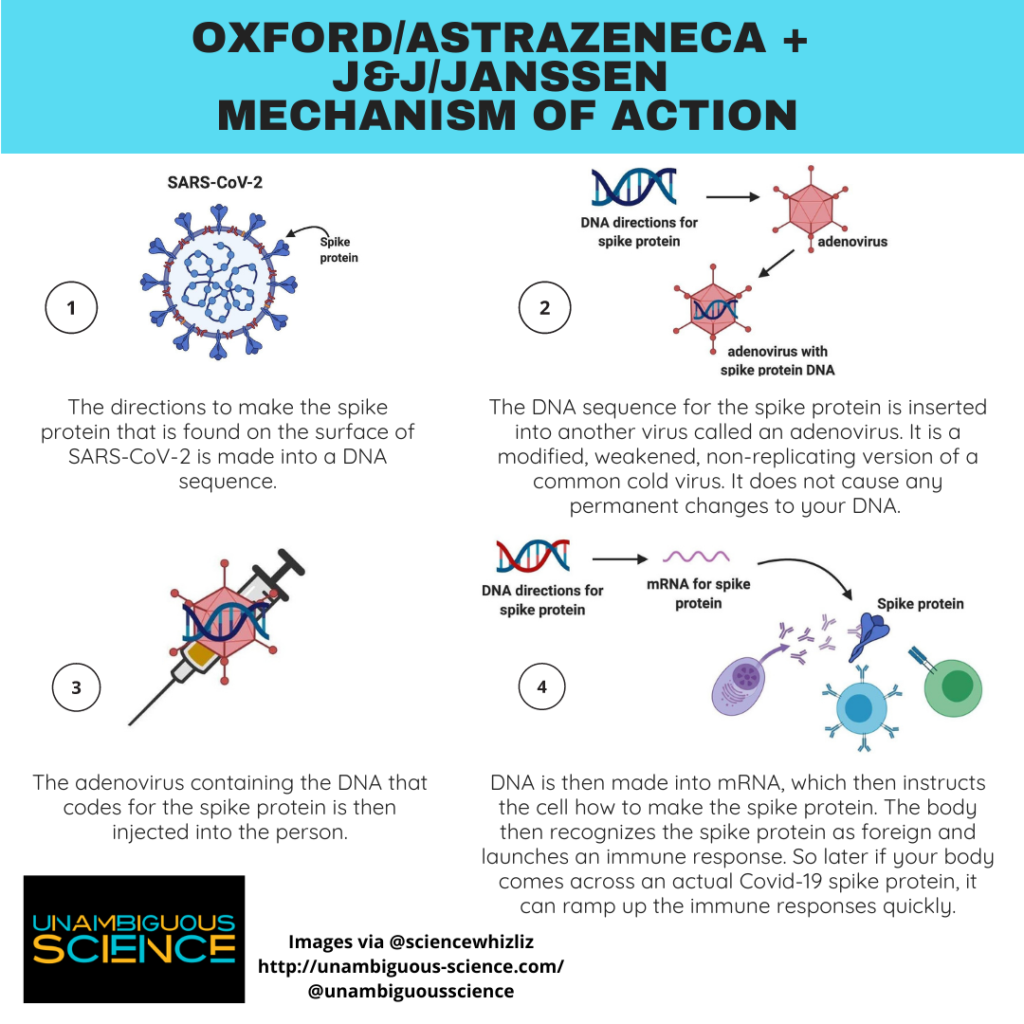
In November 2020, Aspen announced that it had reached an agreement to “perform formulation, filling and secondary packaging” for Johnson & Johnson’s COVID-19 vaccine. What followed, said Nicolaou, was “frenzied activity to try and acquire additional capacity” to enable its facility to make vaccines. Shortly before the pandemic, Aspen had invested around $400 million to expand its sterile capacities and volumes as it planned to relocate the production of some of its general anaesthetics and muscle blockers to Gqeberha.īut when the company saw what was happening with COVID-19, it decided to switch to vaccine production. With the waning of COVID-19, maintaining countries’ and companies’ interest in building regional vaccine manufacturing ability is a challenge – and Aspen’s difficulties should be well noted.Īside from being Africa’s biggest pharmaceutical company, Aspen is also the leading manufacturer and supplier of general anaesthetics in the world outside of the US.Īspen’s main production facility, comprising six different manufacturing units, is in Gqeberha, an economically depressed city in one of the poorest regions in South Africa. But Bangladesh had passed over the vaccine, being made locally by a company called Incepta, in favour of the mRNA vaccines. The Center then affected a technology transfer to Indian and Indonesian manufacturers that went on to make over 100 million doses, he told the seminar. Hotez, who directs the Texas Children’s Hospital Center For Vaccine Development, assisted to develop a viral vector vaccine concept. When that was eventually resolved, the world had fallen for mRNA vaccines and no longer wanted the viral vector vaccine that Aspen had been licensed to produce by Johnson & Johnson. Then Aspen became caught in a political stand-off between Africa and Europe about the fate of African-produced vaccines which delayed production by months. First, despite the hype about the need for African-produced vaccines, African governments failed to buy the locally-made vaccines. Three key factors combined to undermine Aspen’s vaccines. While Nicolaou’s comment sounds mild, the body blow that his company took would have bankrupted a smaller company. “Unless there is security around domestic or regional procurement, you’re going to be very guarded about getting into this business ever again,” Aspen’s Stavros Nicolaou, head of Strategic Trade, told a recent webinar hosted by Brown University’s Professor Wilmot James.  
While the pandemic accord currently being negotiated is almost certain to support regional vaccine production, setting this up is complex and the COVID-era failures offer a number of sobering and cautionary lessons. Meanwhile, in a “demoralising blow”, Bangladesh’s government rejected a vaccine developed by Professor Peter Hotez that was being made by a local company because it wasn’t an mRNA vaccine. Regional vaccine production features in the draft pandemic accord, but there is still a long road before this becomes a realityĪspen Pharmacare invested millions of dollars in scaling up its South African production plant to make COVID-19 vaccines – yet it never sold a single vial. South African President Cyril Ramaphosa visits Aspen Pharmacare’s manufacturing facility in Gqeberha. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
